Rationale. Acute lymphoblastic leukemia (ALL) with unfavorable biology and/or high minimal residual disease (MRD) levels are at high-risk of disease recurrence. Based on the results of the GRAALL-2005/T study (Trinquand A et al, JCO 2013; Beldjord K et al, Blood 2014), T-ALL patients enrolled in the GRAALL-2014/T trial were classified at high-risk (HR) if presenting an unfavorable 4-gene classifier (absence of NOTCH1/FBXW7 mutations or presence of RAS/PTEN alterations) and/or a post-induction IG/TR MRD1 ≥ 10 -4 (including the need of a salvage course to reach a complete remission [CR]). In the phase 2 ATRIALL sub-study, HR patients were offered to receive nelarabine (NELA) during consolidation and maintenance or as bridge to allogeneic hematopoietic stem cell transplantation (HSCT). The aim of the present report is to compare their outcome to that of similar patients treated in the same GRAALL-2014/T trial but without frontline NELA.
Method. Between November 2015 and May 2022, the GRAALL-2014/T included 325 patients with T-ALL. Among these patients, 199 presented with HR features. The ATRIALL sub-study was activated in April 2017. ATRIALL eligibility criteria were defined as HR features, no CNS involvement at baseline, and continuous CR before to start consolidation 2 (first NELA course) at week 12. Based on the results of the COG phase 3 study, the protocol was amended in January 2021 to include patients with CNS involvement at baseline. NELA was given at 1,500 mg/sqm/day at day 1,3 and 5 in combination with etoposide and cyclophosphamide for a maximum of 5 courses during consolidation (2 cycles) and maintenance (3 cycles). Patients with MRD1 ≥ 10 -3 and/or with post-consolidation 1 MRD2 ≥ 10 -4 were eligible for HSCT after one NELA course. A total of 121 patients were included. One patient was excluded due to overt relapse before to start NELA. Eight patients had CNS involvement at baseline. A total of 33 patients included in the GRAALL-2014/T met initial ATRIALL criteria but were not included in the sub-study either before ATRIALL activation (N=21), or during accrual (N=10), or after accrual completion (N=2) and received standard of care (SOC) without NELA. The ATRIALL/CNS- cohort (N=112) was compared to the control cohort (N=33) for MRD response, cumulative incidence of relapse (CIR), disease-free survival (DFS) and overall survival (OS).
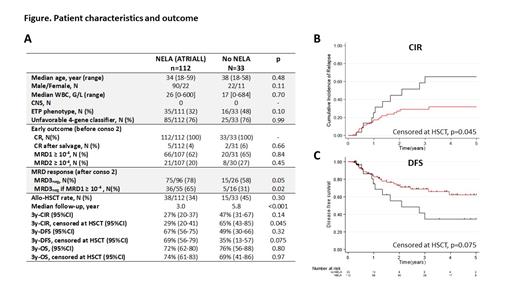
Results. The median follow-up in the ATRIALL study (n=120) was 2.9 years (95%CI, 2.5-3.5). At 3 years, the CIR, DFS, and OS were 28% (95%CI, 20-37), 67% (95%CI, 55-76), and 71% (95%CI, 61-79), respectively. Patients from the ATRIALL/CNS- (N=112) and control (N=33) cohorts exhibited similar characteristics, except for a longer median follow-up in the control cohort (Figure A). Patients included in the ATRIALL study reached lower post-consolidation MRD3 levels (p=0.05). Among patients with MRD1 ≥10 -4, undetectable MRD3 was achieved in 36/55 (65%) after NELA versus 5/16 (31%) after SOC (p=0.02). When censoring at HSCT time, NELA was associated with a significantly reduced CIR (SHR=0.48; 95%CI, 0.23-0.99; p=0.045) and a trend for a prolonged DFS (HR=0.52; 95%CI, 0.26-1.07; p=0.075) (Figure B,C). This difference did not reach significance when patients were not censored at HSCT time. Indeed, high-risk patients not eligible for HSCT benefited from NELA in terms of CIR (SHR=0.43; 95%CI, 0.20-0.98; p=0.045) and DFS (HR=0.46; 95%CI, 0.21-1.03; p=0.06). Patients with non-ETP ALL had a significantly improved CIR (SHR=0.03; 95%CI, 0.17-0.89; p=0.025) and DFS (HR=0.47; 95%CI, 0.22-0.99; p=0.048), whereas ETP-ALL patients did not benefit from NELA over SOC. There was no benefit of prior NELA in patients who received alloHSCT per protocol.
Conclusion. While NELA did not yield an overall improved outcome in the study population, benefits were observed in favorable MRD responders and non-ETP patients. Additional prospective studies are needed to further delineate the specific patient subgroups that might benefit from NELA.
OffLabel Disclosure:
Boissel:ARIAD/Incyte: Honoraria; Astellas Pharma: Honoraria; Servier: Consultancy, Honoraria, Other: Advisory role; Amgen: Consultancy, Honoraria, Other: Expert Testimony and advisory role, Research Funding; Novartis: Consultancy, Honoraria, Other: Advisory role, Research Funding. Huguet:NOVARTIS: Honoraria; INCYTE: Honoraria; PFIZER: Honoraria. Cluzeau:Novartis: Consultancy, Speakers Bureau; Incyte: Speakers Bureau; BMS: Consultancy, Speakers Bureau; Keros: Speakers Bureau; Syros: Speakers Bureau; Jazz Pharma: Consultancy, Speakers Bureau; Abbvie: Consultancy, Speakers Bureau; Servier: Consultancy, Speakers Bureau. Chevallier:Mallinckrodt Pharmaceuticals: Honoraria; Sanofi: Honoraria; Incyte: Honoraria, Research Funding; Takeda: Honoraria; Immedica Pharma: Honoraria; Servier: Honoraria. Jourdan:Abbvie: Honoraria, Membership on an entity's Board of Directors or advisory committees; BMS: Honoraria, Membership on an entity's Board of Directors or advisory committees; Novartis: Honoraria, Membership on an entity's Board of Directors or advisory committees; GSK: Honoraria, Membership on an entity's Board of Directors or advisory committees. Chalandon:Servier: Honoraria; Sanofi: Other: travel support; Janssen: Other: travel support; Astra-Zeneca: Honoraria, Other: travel support; Amgen: Honoraria, Other: travel support; Gilead: Honoraria, Other: travel support; Jazz: Honoraria, Other: travel support, Speakers Bureau; Roche: Honoraria, Other: travel support; Abbvie: Honoraria, Other: travel support; Pfizer: Honoraria; Novartis: Honoraria, Other: travel support; Incyte: Honoraria, Other: travel support; MSD: Honoraria, Other: travel support; BMS: Honoraria, Other: travel support. Dombret:Pfizer: Research Funding; Servier: Membership on an entity's Board of Directors or advisory committees, Research Funding; Astellas: Research Funding; Jazz Pharmaceuticals: Membership on an entity's Board of Directors or advisory committees, Research Funding; Incyte: Membership on an entity's Board of Directors or advisory committees.
Nelarabine frontline

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal